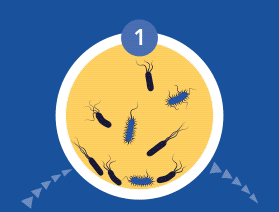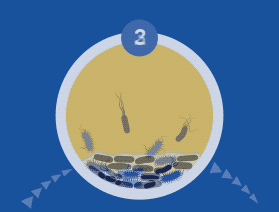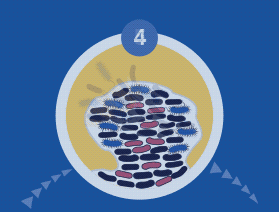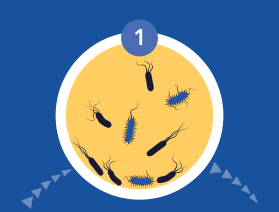
60% of Chronic Wounds Contain Biofilms
Planktonic vs. biofilm phenotypes
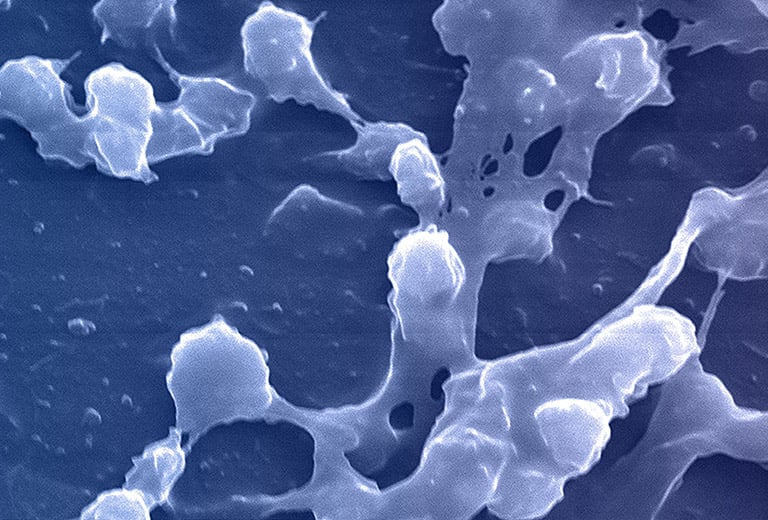
All microorganisms can exist in both the planktonic (free-living) and biofilm phenotypic states, but tend to gravitate towards a biofilm phenotype due to their preference for attaching to each other and to surfaces. Biofilms begin forming within 24 hours, and clinically should be assumed to be present in all chronic wounds. This is a significant cause of wound healing delay, leading to chronicity. In addition, the biofilm mode of growth drastically reduces susceptibility to all types of antimicrobial agents.
Debridement of a biofilm opens a time-dependent window (2-3 days in wounds) where topical treatment agents are more effective and suppress biofilm regrowth. A comprehensive pathologic biofilm view from NGS allows application of specific topical antibiotics and biocides that begin the biofilm reduction process. Note that PCR can only identify organisms for which the individual organism’s PCR probe is used, significantly limiting the organisms identified — and likely missing the “pathologic biofilm” picture. In addition, PCR panels rarely target fungus beyond Candida albicans.
According to the 2017 “Consensus guidelines for the identification and treatment of biofilms in chronic nonhealing wounds:”
- Biofilms are extremely tolerant to multiple forms of antimicrobial treatment
- Systemic antibiotics cannot eradicate a wound biofilm
- Topical appropriate antiseptics are recommended as first‐line therapy in stalled wounds
Biofilms present multiple mechanisms for disrupting wound healing
Elevated proteases present in wound biofilms are associated with destruction of important extracellular matrix (EPS) proteins, destruction of important growth factors, and degradation of key membrane receptor proteins for growth factors.
Additional hypothesized mechanisms in biofilms that interfere with wound healing include:
- Promotion of granulation tissue formation
- Depletion of wound-site oxygen
- Sustained hyper-inflation and host tissue degradation
Disrupting biofilm formation is foundational in infection and inflammation management for chronic wounds. However, complete removal of a biofilm through debridement is unlikely due to perivascular spread below the surface of the wounds. Identifying the multiple bacteria and fungi that make up a specific biofilm using NGS allows direct therapeutic maneuvers such as application of specific topical antibiotics and biocides to increase the effectiveness of debridement.
“In patients who received personalized topical therapeutic gels, based on the results of molecular diagnostics, the average healing time and healing rate reduced significantly and reliance on systemic antibiotics was nearly eliminated. We believe these results will have significant ramifications for wound management.”
– Randall D. Wolcott, MD, Wound & Burn Care Specialist
Read Molecular Diagnostics for Wound Care study
References
- James, G. et al. Biofilms in chronic wounds. Wound Repair Regen. Jan-Feb 2008;16(1):37-44. DOI: 1111/j.1524-475X.2007.00321.x
- Schultz, G. et al. Consensus guidelines for the identification and treatment of biofilms in chronic nonhealing wounds. Wound Repair and Regeneration, September 2018; https://doi.org/10.1111/wrr.12590
- Metcalf, D. and Bowler, P., Biofilm delays wound healing: A review of the evidence. Burns and Trauma, June 2015. 4103/2321-3868.113329
- Dowd, S.E. et al Molecular Diagnostics and personalized medicine in wound care: assessment of outcomes. Journal of Wound Care. 2011; 20: 5, 232-239
- Jo M Martin1, Jonathan M Zenilman, Gerald S Lazarus. Molecular microbiology: new dimensions for cutaneous biology and wound healing. J of invest Dermatol. 2010 Jan;130(1):38-48. doi: 10.1038/jid.2009.221.
- Christopher Attinger and Randy Wolcott. Clinically Addressing Biofilm in Chronic Wounds. Adv Wound Care (New Rochelle). 2012 Jun; 1(3): 127–132. doi: 1089/wound.2011.0333
- PCR panels rarely target fungus beyond Candida albicans. — competitive info: data on file.