In Sequence: The Unseen Commitment to Quality at MicroGenDX
When you receive your test results from MicroGenDX, you are seeing the final product of a comprehensive lab quality journey that began long before your sample arrived at our laboratory. What you don’t see are the countless hours of validation, the daily quality checks, the rigorous training programs, and the unwavering commitment to accuracy that happens behind our laboratory doors.
Let’s pull back the curtain and show you what really goes on in our lab.
The Foundation: Test Validation and Development
Before any of our tests are approved for real world use and begin running patient samples, they have undergone an exhaustive validation process that can take weeks or even months to complete. At MicroGenDX, we don’t take shortcuts when it comes to ensuring the molecular assays behind our tests are ready for clinical use.
Our traditional PCR-based assays follow a methodical nine-step process: primer and probe design, primer concentration optimization, probe concentration optimization, multiplex assay optimization, sensitivity determination with reproducibility data, cycle threshold (Ct) value cutoff establishment, specificity and cross-reactivity testing, precision testing across different technicians and runs, and finally comparison against alternative methods with discrepancy resolution. Each step builds upon the last, creating a cascade of precision that must be perfected before we move forward.
Our Laboratory Director sets acceptable criteria at ≥95% or better for accuracy, sensitivity, and specificity. For precision, we demand 100% reproducibility. The limit of detection must be ≥95% reproducible.
Next-Generation Sequencing: Raising the Bar Even Higher
Our targeted Next-Generation Sequencing (tNGS) assays require an even more comprehensive approach. For bacterial and fungal tNGS, we maintain an average of 7500X and 5500X coverage, respectively. Think of coverage depth like taking multiple photographs of the same scene from slightly different angles. The more images you have, the more confident you can be about what you’re actually seeing.
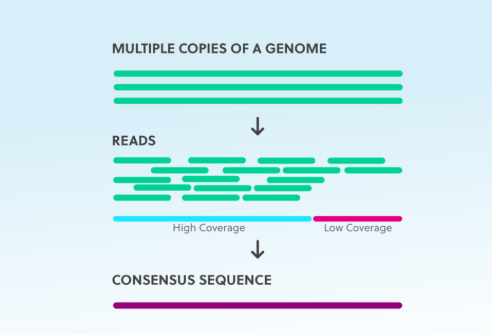
Analytical performance must be established for every aspect of the assay including sample type and collection, extraction procedures, library preparation, data analysis, and result interpretation. The validation must include demonstration of the accuracy and reproducibility of the bioinformatic process as well. For specificity testing, we evaluate many organisms, including genetically related organisms that could be isolated from the original specimen. Inter-assay and Intra-assay reproducibility requires multiple isolates analyzed in triplicate at or near the limit of detection on each of three different days, on different analyzers, by different technicians. For accuracy validation, we test at least thirty isolates representing a wide range of potentially reportable results in a blinded and randomized study.
The Daily Discipline: Quality Control in Action
While test validation happens before patient even testing begins, quality control is an ongoing commitment that occurs every single day our laboratory operates.
Every morning, before any patient sample is tested, our laboratory technicians run a series of controls that verify our systems are performing as expected. For our PCR assays, this means running an internal control on every sample to verify that successful DNA extraction occurred. A negative extraction control runs daily to catch any contamination. For qualitative assays, we perform two external amplification controls daily: a positive control and a negative control, run on all duplicate instruments used that day. For quantitative assays, we elevate this to three external controls: high positive, low positive, and negative.
Before any patient results leave our laboratory, every control is reviewed by our Lab Director, their designee, or trained technicians. This review process creates a checkpoint that catches problems before they reach physicians or patients. Scheduled reviews allow timely detection of trends that might indicate analyzer, control, or reagent problems, enabling resolution before these issues compromise patient samples.
> Consider adding a small section on what we do if a control fails or some kind of issue is detected.
Proving Our Performance: Proficiency Testing
Beyond our internal quality control measures, we participate in external proficiency testing that compares our performance against other laboratories nationwide. Proficiency testing programs periodically send multiple specimens to laboratories across the country for analysis and identification, then compare each laboratory’s results with those of other laboratories and with assigned reference values.
Let’s just say we take proficiency testing a bit more seriously than certain silicon-valley-black-turtleneck-wearing operations that shall remain nameless (though their spectacular implosion did make for excellent HBO viewing). Unlike laboratories that might have been faking their results (hypothetically speaking, of course) or using commercially available equipment when they claimed to have revolutionary technology, we actually perform the tests we say we perform, using the methods we say we use, and achieve the results we report.
And speaking of those results: MicroGenDX maintains a 99.2% accuracy rate with third-party verification across over 1,000 blinded specimens spanning multiple proficiency testing panels over multiple years. Let that sink in for a moment. When we test a sample that’s been anonymized and sent to us alongside hundreds of other laboratories for external evaluation, we get it right 99.2% of the time. We are talking 99.2% verified accuracy across respiratory viruses, methicillin-resistant Staphylococcus aureus, fungi, Group B Streptococcus, vancomycin-resistant Enterococcus, gastrointestinal pathogens, Chlamydia trachomatisand Neisseria gonorrhoeae, Trichomonas vaginalis, and alternate proficiency samples.
Some of these panels we have been crushing with perfect 100% scores for over a decade. Why not 100% on everything? Because science is honest about its limitations, and occasionally a sample is so degraded, so unusual, or so challenging that even the world’s best laboratories might interpret it differently. What matters is that we are transparent about these rare discrepancies, we investigate them thoroughly, and we use them as learning opportunities.
At MicroGenDX, we integrate proficiency testing samples into our routine laboratory workflow, ensuring that these external assessments reflect our actual day-to-day performance rather than idealized conditions. Different employees rotate through proficiency testing evaluation, providing a comprehensive view of our entire team’s competency rather than showcasing only our most experienced technicians.
Unlike certain operations that allegedly showed investors fake results while using commercially available machines behind the scenes, our proficiency testing scores are public, verifiable, and reviewed by regulatory authorities. Revolutionary? Maybe not. Reliable? Absolutely.
The Human Element: Training and Competency
Technology and protocols matter, but at the heart of every accurate test result is a skilled, well-trained laboratory technician.
The qualities we look for before they can even begin training? Our lab techs typically come from the Molecular Pathology Master’s Program at Texas Tech where they are trained on clinical molecular testing methods. We also accept technicians with bachelor’s degrees and experience. CAP has specific qualifications for testing personnel.
Our competency assessment program begins before any new employee even touches a real patient sample. Initial training includes both written examinations and observed competency assessment. But training doesn’t stop after the first few weeks. We conduct a six-month assessment that includes observed competencies for all standard operating procedures related to patient testing. Annual assessments follow thereafter, with direct observation of test performance.
Beyond formal competency assessments, we require continuing education to ensure our staff stays current with evolving technologies and best practices. Each laboratory employee earns a minimum of 12 continuing education units (CEUs) per year through seminars, in-services, work-related reading, vendor training, laboratory-required classes, and participation on facility accrediting inspection teams not only to maintain their professional certifications through ASCP or other certifying bodies but also to comply with the laboratory’s self-imposed standard of excellence.
The Regulatory Reality: CAP, CLIA, CLEP, and Why the FDA (Still) Doesn’t Regulate Us
All of the quality measures described above aren’t optional practices. They’re requirements mandated by the regulatory bodies that oversee clinical laboratories in the United States. MicroGenDX operates under accreditation from the College of American Pathologists (CAP) and certification under the Clinical Laboratory Improvement Amendments (CLIA), and we submit our NGS assays to the New York State Department of Health’s Clinical Laboratory Evaluation Program (CLEP).
Here’s where things get interesting: unlike many diagnostic companies you might see advertising on television, MicroGenDX is not regulated by the Food and Drug Administration. So, you may be wondering: Why is MicroGenDX not regulated by the FDA? Because we are a clinical reference laboratory running Laboratory Developed Tests (LDTs) that we have validated in-house for use in our own laboratory. We’re not manufacturing test kits and selling them to hospitals and laboratories around the world.
Think of it like the difference between a restaurant and a grocery store. A grocery store manufactures or packages food products, puts them in boxes with labels and instructions, and ships them to thousands of retail locations. That grocery store needs FDA oversight because their products are going everywhere, and a problem could affect millions of people across the country. A restaurant, on the other hand, prepares food on-site using recipes and methods they’ve developed, serves it directly to customers who trust the chef’s expertise, and is regulated by local health departments and state agencies that inspect the kitchen, verify the chef’s training, and ensure food safety standards are met.
MicroGenDX is the restaurant in this analogy. We develop our tests, validate them extensively, run them in our own laboratory under direct oversight, and report results directly to physicians who trust our expertise. We’re regulated by CAP (our accrediting body), CLIA (federal certification), and CLEP (state approval for complex molecular tests).
For years, the FDA attempted to gain regulatory authority over LDTs, arguing that all diagnostic tests should fall under their purview regardless of whether they are manufactured test kits or laboratory services. The courts struck down these efforts, recognizing that LDTs are not the same as test systems sold globally and require a different regulatory approach. The legal system understood what some folks at the FDA initially didn’t: the existing regulatory framework—when properly enforced—provides robust oversight of laboratory services without imposing manufacturing regulations on laboratory testing.
CAP accreditation represents the gold standard in laboratory quality. To achieve and maintain CAP accreditation, our laboratory must pass comprehensive inspections that examine every aspect of our operations. CAP inspectors are themselves laboratory professionals who understand the complexities of molecular diagnostics and hold us to the highest possible standards.
The New York State CLEP program takes oversight even further. When we developed our NGS testing capabilities, we submitted comprehensive validation packages to CLEP reviewers—PhD scientists and laboratory directors with deep expertise in molecular microbiology—who scrutinized every aspect of our validation studies, standard operating procedures, quality control protocols, and bioinformatic pipelines. CLEP approval isn’t a rubber stamp. It’s a rigorous scientific review by experts who understand molecular diagnostics at the deepest technical level.
Together, CAP accreditation, CLIA certification, and CLEP approval provide external validation that our internal quality systems meet or exceed national standards. When you receive results from a CAP-accredited, CLIA-certified, CLEP-approved laboratory like MicroGenDX, you’re receiving results from a facility that has been independently verified to meet the highest quality standards in laboratory medicine.
The Unseen Difference
What you have read here represents just a fraction of the quality infrastructure supporting every test result that leaves our laboratory. Behind every bacterial identification, every antibiotic resistance pattern, and every negative result is a comprehensive system designed to ensure accuracy, reliability, and clinical utility.
This commitment to quality is expensive in both time and resources. It would be much easier to run one-size fits all FDA-approved assays. But at MicroGenDX, we recognize that easier isn’t always better when patient care hangs in the balance. Physicians make treatment decisions based on our results. Patients take antibiotics or undergo procedures guided by what we report.
The next time you receive a test result from MicroGenDX, remember that what you are seeing is just the visible tip of an enormous quality iceberg. Below the surface lies the test validation that confirmed our assays work as intended, the daily quality controls that verified our instruments performed correctly, the proficiency testing that compared our results against national standards (and gave us a 99.2% accuracy rate that we’re pretty proud of), the training and competency assessments that ensured qualified personnel performed your test, and the accreditation and certification that provide external validation of our entire quality system.

Quality in laboratory medicine isn’t about flashy marketing or impressive technology alone. It’s about the daily discipline of doing things right, even when no one is watching. It’s about catching errors before they reach patient reports. It’s about continuous improvement and never being satisfied with “good enough.”
At MicroGenDX, this commitment to quality isn’t just what we do; it’s who we are. And while you may never see the quality control logs, the competency assessment forms, or the proficiency testing results, you can trust that they’re there, working behind the scenes to ensure that your results are accurate, reliable, and worthy of the trust you and your physician place in them.
Because in the end, quality isn’t about us. It’s about you, the patient or healthcare provider, receiving the accurate information you need to get better or help others get better. And that’s a commitment worth every hour of validation testing, every daily control run, every training session, every regulatory inspection, and every blinded proficiency sample we undergo.
That’s the unseen difference that makes all the difference.



